 The analysis technique applied is the descriptive analysis and the average analysis techniques. 
The instruments used in this study were a validation test questionnaire and a student response questionnaire. The research method used in this research is the research development method with the development model of ADDIE (Analysis, Design, Development, Implementation and Evaluation). This study aims to develop a physics calculator application on the subject of thermodynamics using Matlab software. Currently, Matlab can be used as a medium for learning physics by students to understand the material well. Matlab is usually used in numerical analysis. It is necessary to have a learning media to be able to visualize the thermodynamics material. You can also select the units (if any) for Input(s) and the Output as well.In the material of thermodynamics, many physics equations make students abstract in understanding the material. With our tool, you need to enter the respective value for Change in Internal Energy & Heat Transferred in Thermodynamic Process and hit the calculate button. To calculate Work using First Law of Thermodynamics, you need Change in Internal Energy (ΔU) & Heat Transferred in Thermodynamic Process (Q). Energy can also transfer from surroundings to system in a sign convention used in physics, such work has a negative magnitude is calculated using Work done in Thermodynamic Process = Change in Internal Energy- Heat Transferred in Thermodynamic Process. in surroundings, through suitable passive linkages, work can lift a weight, for example. in thermodynamics, work performed by a system is energy transferred by system to its surroundings, by a mechanism through which system can spontaneously exert macroscopic forces on its surroundings. The Work using First Law of thermodynamics formula is defined as difference between internal energy and heat into system. It is the energy necessary to create or prepare the system in any given internal state & Heat Transferred in Thermodynamic Process is the form of energy that is transferred from the high-temperature system to the low-temperature system. The Change in Internal Energy of a thermodynamic system is the energy contained within it. Energy can also transfer from surroundings to system in a sign convention used in physics, such work has a negative magnitude and is represented as W = ΔU- Q or Work done in Thermodynamic Process = Change in Internal Energy- Heat Transferred in Thermodynamic Process. Here is how the Work using First Law of Thermodynamics calculation can be explained with given input values -> 300 = 900-600. How to calculate Work using First Law of Thermodynamics using this online calculator? To use this online calculator for Work using First Law of Thermodynamics, enter Change in Internal Energy (ΔU) & Heat Transferred in Thermodynamic Process (Q) and hit the calculate button. 
Work done in Thermodynamic Process is denoted by W symbol. Energy can also transfer from surroundings to system in a sign convention used in physics, such work has a negative magnitude. 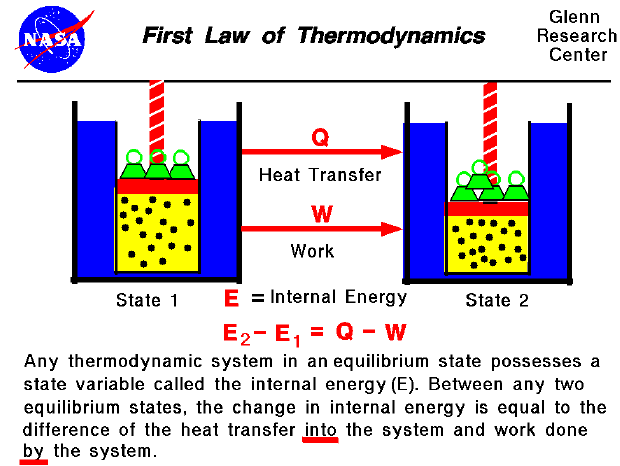
Work using First Law of Thermodynamics calculator uses Work done in Thermodynamic Process = Change in Internal Energy- Heat Transferred in Thermodynamic Process to calculate the Work done in Thermodynamic Process, The Work using First Law of thermodynamics formula is defined as difference between internal energy and heat into system. How to Calculate Work using First Law of Thermodynamics? 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
